
The Bjerrum plot shows typical equilibrium concentrations, in solution, in seawater, of carbon dioxide and the various species derived from it, as a function of pH. What is the precipitate formed between the reaction of carbonic acid and sodium hydroxide H2CO3 + 2 NaOH Na2CO3 + 2 H2O sodium. It remains only on protected surfaces that are not directly washed by the rain. Equilibrium constant values Bjerrum plot for carbonate speciation in seawater (ionic strength 0.7 mol dm 3) In aqueous solution carbonic acid behaves as a dibasic acid. Calcium in a sulfate form is soluble in water however, calcium sulfate (gypsum) can precipitate at high rates. You generally get a fine precipitate of CaCO3(s), the which you might not. Gypsum is soluble in water although it can form anywhere on carbonate stone surfaces that are exposed to sulfur dioxide gas (SO2), it is usually washed away. CO2(g) + Ca(OH)2(aq)rarrCaCO3(s)darr +H2O(l) The reaction is NOT so obvious. This black crust is primarily composed of gypsum, a mineral that forms from the reaction between calcite, water, and sulfuric acid. However, sheltered areas on limestone and marble buildings and monuments show blackened crusts that have peeled off in some places, revealing crumbling stone beneath. This equation follows from the law of mass action, if it is assumed that at 5 5 to pH 6 the equation C02 -j- H20 H2C03 is catalysed by hydrogen ions. Both compounds are white solids that can be dissolved in water to make clear, colorless solutions. One example is the reaction between lead (II) nitrate and potassium iodide. Report to the correct significant figures. The results at pH5 5 to pH 6, could not be fitted by Faurholt’s equation I, but required, instead, the equation co.C02 HI + H2C03 H. A precipitation reaction is when two aqueous ionic compounds form a new ionic compound that is not soluble in water. Calculate the mass of precipitate that can be formed when starting with 25.48 mL of 0.067 M carbonic acid. This weak carbonic acid reacts with carbonates2 in rocks and silicates3 to form soluble calcium, magnesium and manganese ions4 and other minerals, the. This happens as the carbon dioxide forms acidic carbonic acid when it dissolves in the water, the carbonic acid (H2CO3) reacts further with the calcium. You might expect that sheltered areas of stone buildings and monuments would not be affected by acid precipitation. 12.3.0.4pH 12.9.0Phosphoric acid 12.3.0.2Polyprotic acids Experiments 12.3.15Acids with salts 12.3.13Concentrated acids with metals, sulfuric acid with copper 12.3.5.01Copper oxide with sodium hydrogen sulfate 12.3.0.6Ethanoic acid, CH3COOH, acetic acid Hydrochloric acid 12.3.3. Science Chemistry Chemistry questions and answers Write the balanced chemical equation for the reaction between carbonic acid and iron (III) hydroxide. Precipitation reactions are usually represented solely by net ionic equations. A net ionic equation must be balanced on both sides not only in terms of atoms of elements, but also in terms of electric charge. Stone surface material may be lost all over or only in spots that are more reactive. C+ (aq)+ B- (aq) CB (s) The net ionic equation only shows the precipitation reaction. In exposed areas of buildings and statues, we see roughened surfaces, removal of material, and loss of carved details. ice and club soda are added, the precipitate redissolves. Since the mid-19th century, these fuels have been burned on a huge scale to supply the energy needs of our modern industrial society, releasing tens of millions of tons of additional SO 2 into the atmosphere annually.When sulfurous, sulfuric, and nitric acids in polluted air and rain react with the calcite in marble and limestone, the calcite dissolves. This reaction can be reversed, or carbonic acid can decompose into water and carbon dioxide. Carbonic acid is the product of an acid anhydride reaction between carbon dioxide and water. Many coals contain as much as 5%–6% pyrite (FeS 2) by mass, and fuel oils typically contain at least 0.5% sulfur by mass. When they react, the proton from the acid is transferred to the bicarbonate, making the weak acid carbonic acid. In particular, it is found that aluminum, magnesium and iron salts solubilized along with calcium by acid extraction of sludge precipitate as hydroxides at. 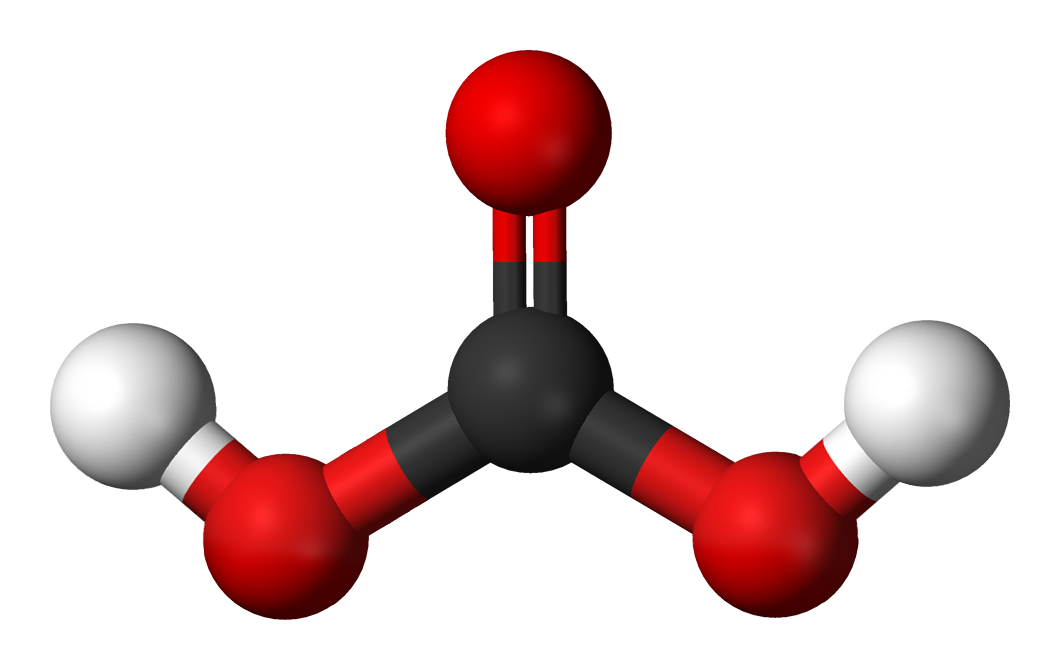
Unfortunately, the burning of fossil fuels seems to have tipped the balance. Large amounts of sulfur dioxide have always been released into the atmosphere by natural sources, such as volcanoes, forest fires, and the microbial decay of organic materials, but for most of Earth’s recorded history the natural cycling of sulfur from the atmosphere into oceans and rocks kept the acidity of rain and snow in check.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
